A .gov website belongs to an official government organization in the United States.
A lock () or https:// means you've safely connected to the .gov website. Share sensitive information only on official, secure websites.

Laboratory studies show that chlorine (Cl) reacts very rapidly with ozone. They also show that the reactive chemical chlorine oxide (ClO) formed in that reaction can undergo further processes which regenerate the original chlorine, allowing the sequence to be repeated very many times (a "chain reaction"). Similar reactions also take place between bromine and ozone.
But do these ozone-destroying reactions occur in the real world? All of our accumulated scientific experience demonstrates that if the conditions of temperature and pressure are like those in the laboratory studies, the same chemical reactions will take place in nature. However, many other reactions including those of other chemical species are often also taking place simultaneously in the stratosphere, making the connections among the changes difficult to untangle. Nevertheless, whenever chlorine (or bromine) and ozone are found together in the stratosphere, the ozone-destroying reactions must be taking place.
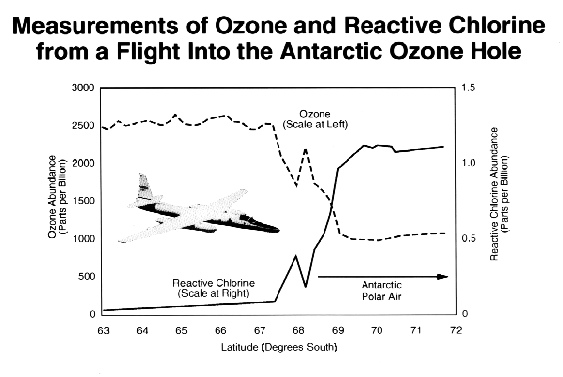
Sometimes a small number of chemical reactions is so important in the natural circumstance that the connections are almost as clear as in laboratory experiments. Such a situation occurs in the Antarctic stratosphere during the springtime formation of the ozone hole. During August and September 1987 - the end of winter and beginning of spring in the Southern Hemisphere - aircraft equipped with many different instruments for measuring a large number of chemical species were flown repeatedly over Antarctica. Among the chemicals measured were ozone and chlorine oxide, the reactive chemical identified in the laboratory as one of the participants in the ozone-destroying chain reactions. On the first flights southward from the southern tip of South America, relatively high concentrations of ozone were measured everywhere over Antarctica. By mid-September, however, the instruments recorded low concentrations of ozone in regions where there were high concentrations of chlorine oxide and vice versa, as shown in the figure. Flights later in September showed even less ozone over Antarctica, as the chlorine continued to react with the stratospheric ozone.
Independent measurements made by these and other instruments on this and other airplanes, from the ground, from balloons, and from satellites have provided a detailed understanding of the chemical reactions going on in the Antarctic stratosphere. Regions with high concentrations of reactive chlorine reach temperatures so cold (less than approximately -80°C, or -112°F) that stratospheric clouds form, a rare occurrence except during the polar winters. These clouds facilitate other chemical reactions that allow the release of chlorine in sunlight. The chemical reactions related to the clouds are now well understood through study under laboratory conditions mimicking those found naturally. Scientists are working to understand the role of such reactions of chlorine and bromine at other latitudes, and the involvement of particles of sulfuric acid from volcanoes or other sources.