A .gov website belongs to an official government organization in the United States.
A lock () or https:// means you've safely connected to the .gov website. Share sensitive information only on official, secure websites.

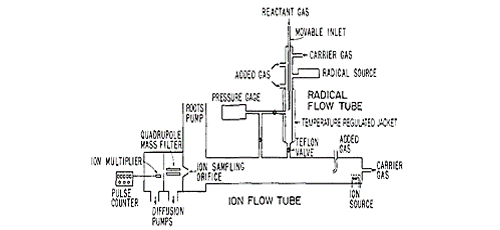
Principle of the Measurement
Chemical ionization relies on the soft ionization of analyte molecules by pre-ionized reactant gas molecules. The collision of these reactant gas fragments and the analyst molecules of interest is tuned to ensure limited fragmentation.
Technical Specifications
Species Measured: small organic molecules of sulfur and nitrogen, halogenated compounds
Detection Limit: ~1 x 109 molecule cm-3, depending on sample
Applications
Absolute temperature and pressure kinetics measurements, gas phase and heterogeneous chemistry: Antarctic ozone hole studies
Key Publications
Longfellow, C.A., A.R. Ravishankara, D.R. Hanson, Reactive and nonreactive uptake on hydrocarbon soot: HNO3, O3, and N2O5, Journal of Geophysical Research, doi:10.1029/2000JD900297, 2000.
Hanson, D. R. Reaction of ClONO2 with H2O and HCl in sulfuric acid and HNO3/H2SO4/H2O mixtures, Journal of Physical Chemistry A, doi:10.1021/jp972767s, 1998.
Hanson, D.R., E.R. Lovejoy, The reaction of ClONO2 with submicrometer sulfuric acid aerosol, Science, doi:10.1126/science.267.5202.1326, 1995.