A .gov website belongs to an official government organization in the United States.
A lock () or https:// means you've safely connected to the .gov website. Share sensitive information only on official, secure websites.


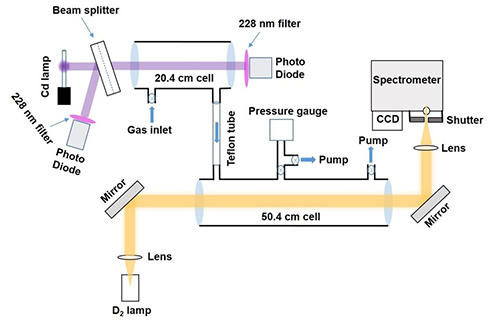
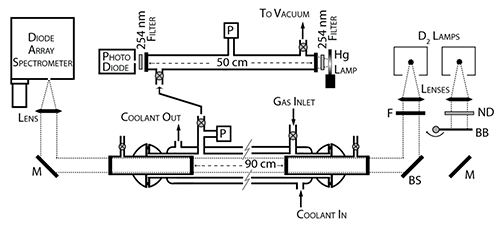
Principle of the Measurement
UV/vis spectroscopy is principally governed by Beer's law, which states that the absorbance of a compound is proportional to the concentration of the compound multiplied by the pathlength of the absorption cell and the UV cross section at a particular wavelength.
Technical Specifications
Bandwidth: ~300 nm
Accuracy: ± 5 x 10-4 absorbance
Wavelength calibration: ± 0.1 nm
Applications
UV spectroscopy: monitoring and quantification of transient species, low volatility compounds, stable species, synthesized samples, and gas concentrations in kinetics measurements
Key Publications
McGillen, M.R., V.C. Papadimitriou, S.C. Smith, and J.B. Burkholder, FC(O)C(O)F, FC(O)CF2C(O)F, and FC(O)CF2CF2C(O)F: Ultraviolet and infrared absorption spectra and 248 nm photolysis products, Journal of Physical Chemistry A, doi:10.1021/acs.jpca.0c04607, 2020.
Papanastasiou, D.K., V.C Papadimitriou, D.W. Fahey, J.B. Burkholder, UV absorption spectrum of the ClO dimer (Cl2O2) between 200 and 420 nm, Journal of Physical Chemistry A, doi:10.1021/jp9065345, 2009.
Marshall, P., V.C. Papadimitriou, D.K. Papanastasiou, J.M. Roberts, and J.B. Burkholder, UV and Infrared absorption spectra and 248 nm photolysis of maleic anhydride (C4H2O3), Journal of Photochemistry and Photobiology A: Chemistry, doi:10.1016/j.jphotochem.2019.111953, 2019.