A .gov website belongs to an official government organization in the United States.
A lock () or https:// means you've safely connected to the .gov website. Share sensitive information only on official, secure websites.
15 March 2011

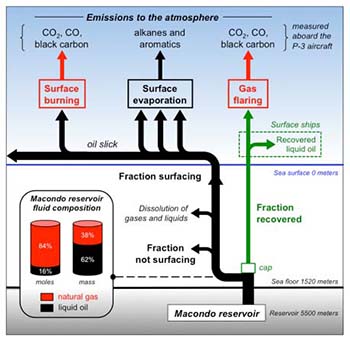
NOAA scientists and academic partners have found a way to use air chemistry measurements taken hundreds of feet above last year's BP Deepwater Horizon oil spill to estimate how fast gases and oil were leaking from the reservoir thousands of feet underwater. The scientists also determined the fate of most of those gas and oil compounds using atmospheric chemistry data collected from the NOAA WP-3D research aircraft overflights in June. The study "Atmospheric emissions from the Deepwater Horizon spill constrain air-water partitioning, hydrocarbon fate, and leak rate" was accepted for publication in Geophysical Research Letters, a publication of the American Geophysical Union.
"We present a new method for understanding the fate of most of the spilled gases and oil," said CSD's Tom Ryerson, lead author of the report. "We found that the spilled gases and oil (spilled fluid) obeyed a simple rule: whether a compound can dissolve or evaporate determines where it goes in the marine environment. That simple rule, and the methods we lay out in this paper, could enable airborne evaluation of the magnitude of future spills."
Knowing where the spilled gas and oil mixture ended up could also help resource managers and others trying to understand environmental exposure levels.
Using the atmospheric measurements and information about the chemical makeup of the leaking reservoir fluid, Ryerson and his colleagues calculated that at least 32,600-47,700 barrels of liquid gases and oil poured out of the breached reservoir on June 10. This range, determined independently of previous estimates presents a lower limit. "Although we accounted for gases that dissolved before reaching the surface, our atmospheric data are essentially blind to gases and oil that remain trapped deep underwater," Ryerson said. Comparison of the new result with official estimates is not possible because this airborne study could not measure that trapped material.
Not including that trapped material, atmospheric measurements combined with reservoir composition information showed that about one-third (by mass) of the oil and gas dissolved into the water column on its way to the surface. The team found another 14 percent by mass (570,000 lbs per day) was lost quickly to the atmosphere within a few hours after surfacing, and an additional 10 percent was lost to the atmosphere over the course of the next 24 to 48 hours.
Among the study's other key findings:
A portion of oil and gas was "recovered" by response activities and piped from the leaking wellhead to the Discoverer Enterprise drill ship on the ocean surface. The research team calculated this recovered fraction by measuring emissions from natural gas flaring aboard the recovery ship. They calculated a recovery rate of 17,400 barrels of reservoir fluid (liquid gas and oil) for June 10, and which accounted for approximately one-third to one-half of the group's total estimate of 32,600-47,700 barrels of fluid per day.
Ryerson and his colleagues concluded that the technique they developed could be applied to future oil spills, whether in shallow or deep water. The Gulf research flights were possible only because a NOAA WP-3D research aircraft had already been outfitted with sensitive chemistry equipment for deployment to California for an air quality and climate study and was redeployed to the Gulf. NOAA's Gulf flights were in support of the Unified Area Command's effort to observe and monitor the environmental effects of the spill.
Thomas B. Ryerson1, Kenneth Aikin1,2, Wayne Angevine1,2, Elliot Atlas3, Donald Blake4, Charles Brock1, Fred Fehsenfeld1,2, Ru-Shan Gao1, Joost de Gouw1,2, David Fahey1, John Holloway1,2, Daniel Lack1,2, Richard Lueb5, Simone Meinardi4, Ann Middlebrook1, Daniel Murphy1, J. Andy Neuman1,2, John Nowak1,2, David Parrish1, Jeff Peischl1,2, Anne Perring1,2, Ilana Pollack1,2, A.R. Ravishankara1, James Roberts1, Joshua Schwarz1,2, J. Ryan Spackman1,2, Harald Stark1,2, Carsten Warneke1,2, and Laurel Watts1,2, Atmospheric emissions from the Deepwater Horizon spill constrain air-water partitioning, hydrocarbon fate, and leak rate, Geophysical Research Letters, doi:10.1029/2011GL046726, 2011.