A .gov website belongs to an official government organization in the United States.
A lock () or https:// means you've safely connected to the .gov website. Share sensitive information only on official, secure websites.
4 June 2013
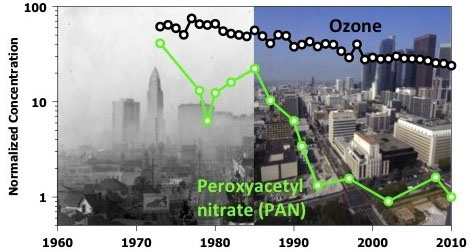
The cleanup of California's tailpipe emissions over the last few decades has not only reduced ozone pollution in the Los Angeles area, it has also altered the pollution chemistry in the atmosphere, making the eye-stinging "organic nitrate" component of air pollution plummet, according to a new study accepted for publication in the Journal of Geophysical Research-Atmospheres, a publication of the American Geophysical Union.
For the study, a team of scientists analyzed new data gathered using a NOAA research aircraft, along with archived data going back a half-century, to produce a comprehensive study of air pollution in the Los Angeles region.
"This is good news: LA's air has lost a lot of its 'sting,'" said lead author Ilana Pollack, a CIRES scientist who works at CSD. "Our study shows exactly how that happened, and confirms that California's policies to control emissions have worked as intended."
Scientists have studied the origins and levels of air pollutants in the South Coast Air Basin – a region encompassing the Los Angeles urban area – for a long time. Since the 1960s, they have measured levels of ozone and other air pollutants that are formed in the atmosphere (so-called "secondary" pollutants) and the ingredients, or "precursors," that form them: volatile organic compounds (VOCs) and nitrogen oxides (NOx). These precursors are directly emitted from various sources, primarily vehicle exhaust in LA but also from power-generating facilities, industry, and natural sources such as vegetation.
As studies began to identify the high levels of air pollution and its causes, policies and controls were implemented to restrict emissions of the NOx and VOC ingredients that result in ozone and other secondary air pollutants. Although the population in the Southern California region has tripled between 1960 and 2010, and the number of vehicles has increased by a similar factor, research studies have indicated that air pollution in the region has decreased - as a result of these policies.
"To most people the big deal is that things have got a lot better," Pollack said. "But as scientists we want to know how they have got better."

To pin down the exact nature of the downward trends and the related changes in the pollution chemistry, Pollack and her team examined new data from research aircraft and archived data from roadside monitors and ground-based instruments. In doing so, they generated a synthesis of information on ozone, other secondary pollutants and pollutant precursors from 1960 to 2010. This work included measurements of ozone and nitrogen oxides collected by Pollack and her colleagues over the South Coast Air Basin using instruments aboard NOAA's WP-3D research aircraft during a California-based field mission in 2010 (CalNex 2010).
The exhaustive approach paid off, and gave the scientists new insights into the changing chemistry of LA's air.
"The emission reductions have 'flipped' some of the chemistry that takes place in the atmosphere," said Pollack. "The relevant precursors in the atmosphere now favor chemical pathways that are more likely to produce nitric acid, and less likely to make ozone and peroxyacetyl nitrate (PAN)."
PAN is the organic nitrate compound historically associated with eye irritation (the 'sting') in Los Angeles smog.
"Compiling long-term trends in precursors and secondary products, then seeing all the data together on paper, really made changes in the chemistry stand out," Pollack said.
The researchers' analysis showed that emission control measures in Southern California have succeeded in driving pollution down even though population has grown, notes Tom Ryerson, NOAA scientist and coauthor of the new study. Motor vehicles remain the dominant source of emissions in Los Angeles.
Understanding the past and present chemistry in the atmosphere that creates air pollution is critical to being able to estimate how much pollution will be formed in future years, Pollack said. The researchers hope that this new insight will provide useful information to the policy makers who will be crafting the next generation of policies aimed at improving air quality in the region.
"Our work aims to interpret the past and present observations, with an eye toward informing future decisions," Pollack said.
Coauthors on the study were NOAA CSD scientists Thomas B. Ryerson, Michael Trainer, James M. Roberts and David D. Parrish, and NOAA CSD / CIRES scientist J. Andrew Neuman.
Pollack, Ilana B., Thomas B. Ryerson, Michael Trainer, J. A. Neuman, James M. Roberts, David D. Parrish, Trends in ozone, its precursors, and related secondary oxidation products in Los Angeles, California: A synthesis of measurements from 1960 to 2010, Journal of Geophysical Research, doi:10.1002/jgrd.50472, 2013.
Decreases in ozone (O3) observed in California's South Coast Air Basin (SoCAB) over the past five decades have resulted from decreases in local emissions of its precursors, nitrogen oxides (NOx = NO + NO2) and volatile organic compounds (VOCs). Ozone precursors have been characterized in the SoCAB with measurements dating back to 1960. Here, we compile an extensive historical data set using measurements in the SoCAB between 1960 and 2010. Faster rates of decrease have occurred in abundances of VOCs (-7.3 ± 0.7 % year-1) than in NOx (-2.6 ± 0.3 % year-1), which have resulted in a decrease in VOC/NOx ratio (-4.8 ± 0.9 % year–1) over time. Trends in the NOx oxidation products peroxyacetyl nitrate (PAN) and nitric acid (HNO3), measured in the SoCAB since 1973, show changes in ozone production chemistry resulting from changes in precursor emissions. Decreases in abundances of PAN (-9.3 ± 1.1 % year-1) and HNO3 (-3.0 ± 0.8 % year-1) reflect trends in VOC and NOx precursors. Enhancement ratios of O3 to (PAN + HNO3) show no detectable trend in ozone production efficiency, while a positive trend in the oxidized fraction of total reactive nitrogen (+2.2 ± 0.5 % year-1) suggests that atmospheric oxidation rates of NOx have increased over time as a result of the emissions changes. Changes in NOx oxidation pathways have increasingly favored production of HNO3, a radical termination product associated with quenching the ozone formation cycle.